NANOSYSTEMS: PHYSICS, CHEMISTRY, MATHEMATICS, 2020, 11 (5), P. 608–614
Synthesis Ni-doped CuO nanorods via Successive Ionic Layer Deposition method and their capacitive performance
A. A. Lobinsky – Saint Petersburg State University, Peterhof, 198504 Saint Petersburg, Russia; lobinsky.a@gmail.com
M.V. Kaneva – Saint Petersburg State University, Peterhof, 198504 Saint Petersburg, Russia
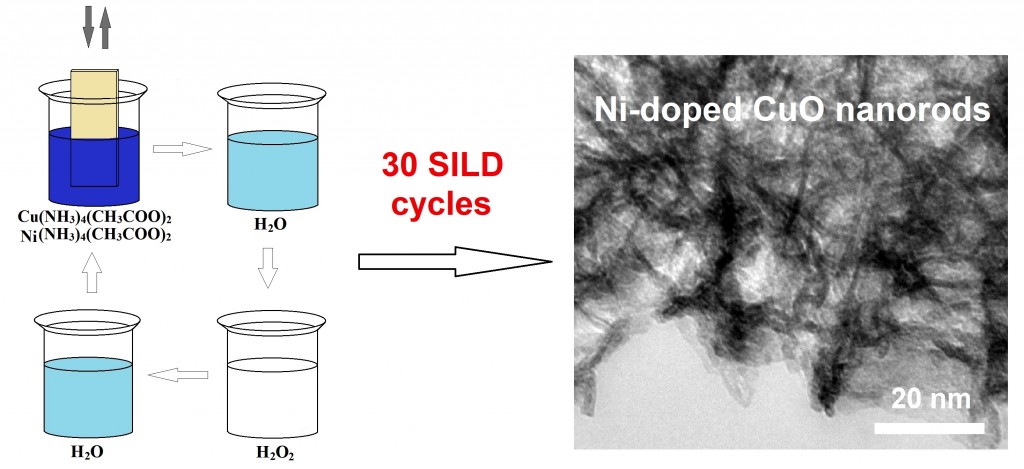
In this work first described the new relatively simple approach to the synthesis of nanolayers of Ni-doped CuO via of Successive Ionic Layer Deposition (SILD) method. The study of Ni-doped CuO nanolayers, synthesized of SILD, has been carried out by HRTEM, XRD, FTIR and XPS spectroscopy methods; it was demonstrated that they had been formed of nanorods with dimensions of about 10–15 nm and tenorite crystal structure CuO were formed. The research electrochemical properties of nanolayers were carried out in 1 KOH solution by using techniques of cyclic voltammetry and galvanostatic curves method. The electrochemical study of nickel foam electrodes modified by Ni-doped CuO nanolayer prepared by 30 SILD cycles demonstrates that specific capacitance is 154 mAh/g (1240 F/g) at current density 1 A/g. Repeated cycling after 1000 charge-discharge cycles demonstrates 8% capacitance fade from the initial value, so such electrodes may be used as effective electroactive materials for alkaline battery and pseudocapacitors.
Keywords: copper oxide, nanocrystal, nanorods, successive ionic layer deposition, electrode materials, alkaline battery.
PACS 81.07.Bc
DOI 10.17586/2220-8054-2020-11-5-608-614