NANOSYSTEMS: PHYSICS, CHEMISTRY, MATHEMATICS, 2021, 12 (2), P. 224–231
The effect of co-precipitation temperature on the crystallite size and aggregation/agglomeration of GdFeO3 nanoparticles
V. I. Popkov – Ioffe Institute, Saint Petersburg, 194021, Russia; vadim.i.popkov@mail.ioffe.ru
Y. Albadi – Saint Petersburg State Institute of Technology, Saint Petersburg, 190013, Russia; Al-Baath University, Homs, 77, Syrian Arab Republic
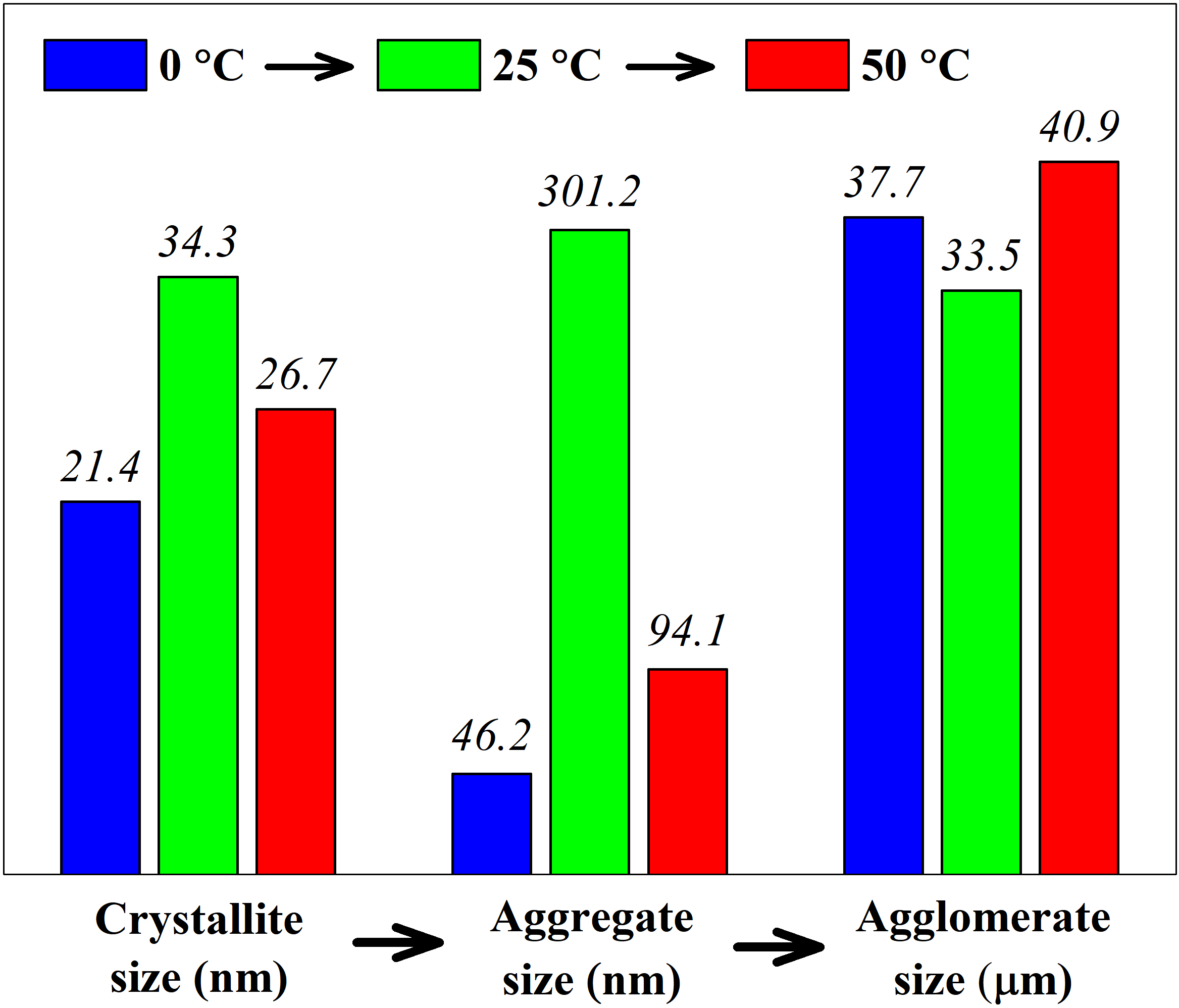
In this work, a series of GdFeO3 nanopowders was successfully synthesized via a reverse co-precipitation technique at different solution temperatures (0, 25 and 50 °C) followed by thermal treatment. Co-precipitated hydroxides and heat treatment products were analyzed using EDXS, DTA-TGA, PXRD, ASA and LD methods. It was shown that the formation temperature of GdFeO3 nanoparticles varies in the range of 737.5–758.8 °C and total weight loss varies in the range of 23.6–26.4% depending on the temperature of initial solutions. The specific surface areas of nanopowders were found to be strongly dependent on the factor mentioned above and belong to 2.5–16.3 m2/g values interval. The hierarchical structure of the obtained nanopowders was established and the effect of co-precipitation temperature on the average crystallite (21.4–34.3 nm), aggregate (46.2–301.2 nm) and agglomerate (33.5–40.9 μm) sizes was discussed in detail.
Keywords: co-precipitation, gadolinium orthoferrite, nanoparticles, aggregation, agglomeration.
PACS 81.20.Fw, 61.46.+w, 75.20.-g
DOI 10.17586/2220-8054-2021-12-2-224-231
[In Russian] В.И. Попков, Ю. Альбади
Влияние температуры соосаждения на размер кристаллитов и агрегацию/агломерацию наночастиц GdFeO3
В данной работе серия нанопорошков GdFeO3 была успешно синтезирована методом обратного соосаждения при различных температурах раствора (0, 25 и 50 °С) с последующей термической обработкой. Соосажденные гидроксиды и продукты термообработки анализировали методами РСМА, ДТА-ТГА, ПРД, АСА и ЛД. Показано, что температура образования наночастиц GdFeO3 колеблется в пределах 737.5–758.8 °С, а общая потеря массы колеблется в пределах 23.6–26.4 % в зависимости от температуры исходных растворов. Установлено, что удельная поверхность нанопорошков сильно зависит от упомянутого выше фактора и находится в интервале значений 2.5–16.3 м2/г. Установлена иерархическая структура полученных нанопорошков и подробно обсуждено влияние температуры соосаждения на средние размеры кристаллитов (21.4–34.3 нм), агрегатов (46.2–301.2 нм) и агломератов (33.5–40.9 мкм).
Ключевые слова: соосаждение, ортоферрит гадолиния, наночастицы, агрегация, агломерация